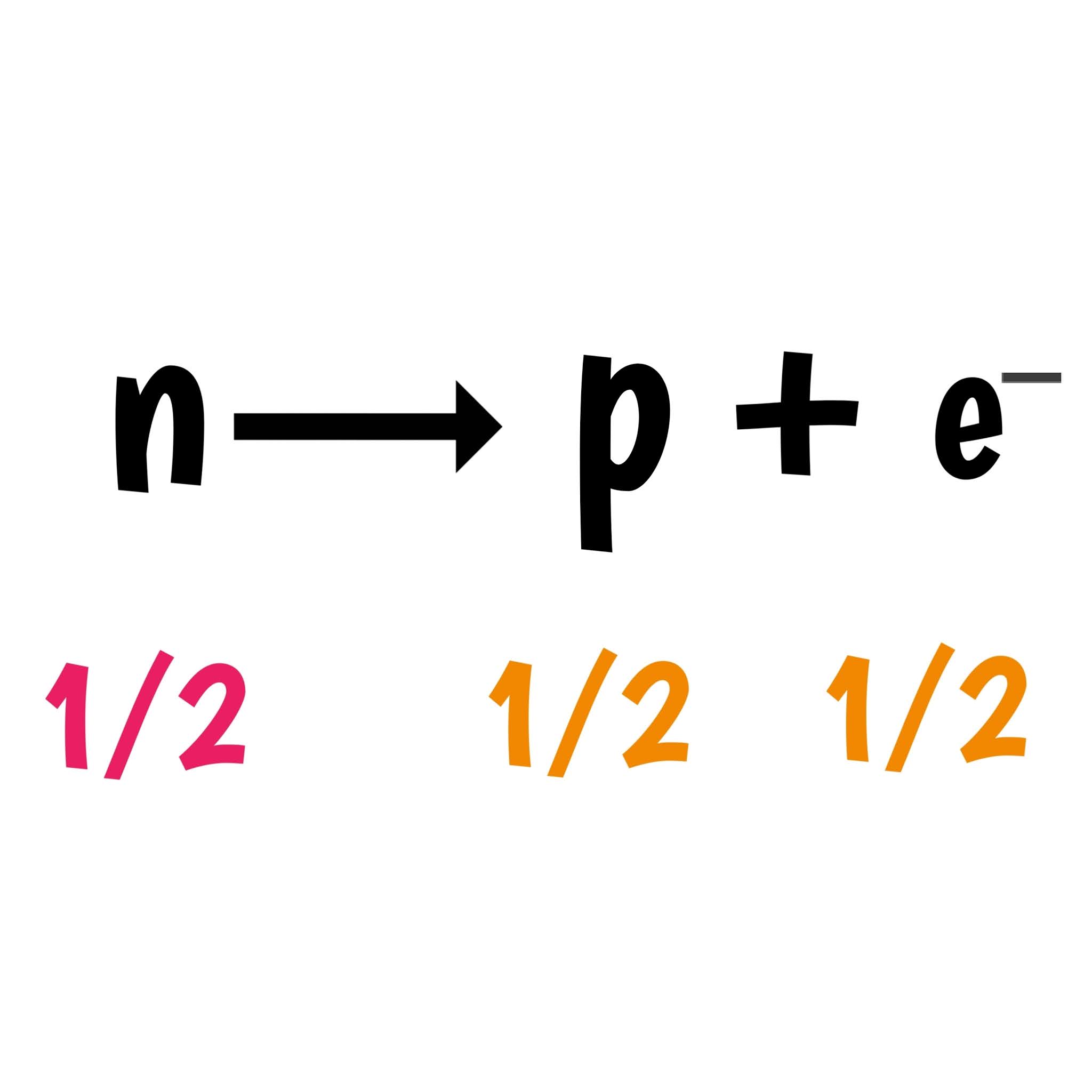
It was expected that the same considerations would hold for a parent nucleus breaking down to a daughter nucleus and a beta particle. In other words, the beam of alpha particles should be monoenergetic. Since the same particles appear as products at every breakdown of a particular parent nucleus, the mass difference should always be the same, and the kinetic energy of the alpha particles should also always be the same. As a result of the law of conservation of energy, this difference appears in the form of the kinetic energy of the alpha particle. For example, in the case of alpha decay, when a parent nucleus breaks down spontaneously to yield a daughter nucleus and an alpha particle, the sum of the mass of the two products does not quite equal the mass of the original nucleus (see Mass Defect). The particle carries the energy from the difference between the initial and final nuclear states. The resulting particle ( alpha particle or photon) has a narrow energy distribution in alpha and gamma decay. The discovery of the neutrino is based on the law of conservation of energy during the process of beta decay. The study of beta decay provided the first physical evidence for the existence of the neutrino. Conversely, a proton is converted into a neutron by the emission of a positron, thus changing the nuclide type. By beta decay emission, a neutron is transformed into a proton by the emission of an electron. High-speed electrons or positrons are emitted during beta decay. Beta radiation consists of high-energy beta particles. Beta-decay (β-decay) is a type of radioactive decay in which a beta particle and a respective neutrino are emitted from an atomic nucleus.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
